Pastry science #2 – The properties of sugars – Part I
In the previous “technical” post, I wrote about the main categories of sugars.
Mono- and disacchararides are called simple sugars, because they are made of only one or two molecules of sugar joined together. All simple sugars share a set of characteristics:
- They are sweet (duh!)
- They give structure
- They form crystals
- They melt
- They absorb humidity
- They preserve
- They lower the freezing point and increase the boiling point of water
- They facilitate the fermentation
- They caramelize and are subject to the Maillard reaction
The Maillard reaction is a big thing in pastry, so big that I also had a question about it in my pastry exam. It is also so big that it deserves another post! To make this article readable, I will only deal with the first 4 points. So, in order…
The sweetness of sugars
We all use sugars instinctively because they are sweet. I am an atypical Italian: I don’t drink coffee, but I know for sure that a coffee without sugar can be a pretty bad experience for many people. Sweetness is anyway subjective (that’s why there are some weird people who drink coffee without sugar by choice! Can you believe that?) and science has found no way to objectively measure with a machine how sweet a sugar is.
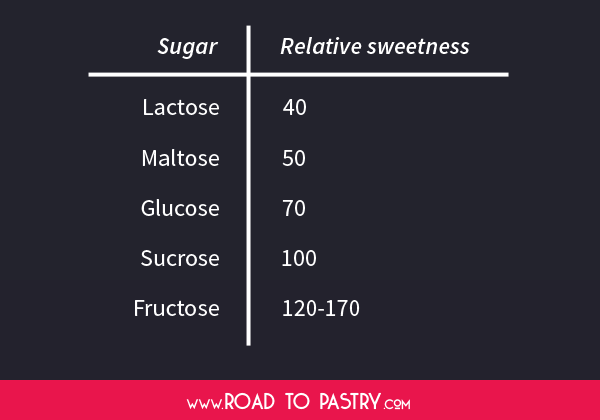
Nevertheless, we can refer to the table below about the “relative sweetness” or “perceived sweetness”, a value obtained after tasting experiments: the value for sucrose was arbitrarily set at 100 and the other sugars were ordered consequently.
For example, from the table we learn that glucose is less sweet than granulated sugar: this means that you’ll need to use a bigger quantity of glucose to have the same sweetness as granulated sugar. Just remember that all sugars have the same amount of calories per gram, though!
Structure in pastry, thanks to sugars
With baking, water evaporates so we must make sure a product has the right structure. Structure can be conferred by gluten, but also by sugar. think of a meringue: it’s just made of egg whites and sugar. A meringue without sugar would collapse.
Sugar can give structure by interfering:
- With the formation of gluten
- With the coagulation of egg proteins
- With the gelatinization of starches
Take another example: a tart dough must be flaky. This means that there must not be too much gluten and not much coagulation of egg proteins. To achieve that result, you need sugar! If you remove too much sugar from the recipe, there will be nothing else preventing the formation of gluten, so your tart will be as hard as a brick (or almost).
On the opposite side, a spongecake with too much sugar could collapse, because the gelatinization of starches has been delayed excessively.
When studying or modifying recipes, keep in mind that different sugars can have different impacts on the structure. For example, sucrose slows down the gelatinization of starches twice as much as fructose.
Crystallization
All simple sugars form crystals. It’s not good or bad per se, you just have to know it and make sure you know what you want for your recipe. Candies or dry baked goods must contain crystallized sugars. A super smooth and shiny cake glaze should definitely not contain crystals.
Here are two tips to prevent the crystallization:
- Mix the main sugar (e.g. sucrose) with around 5-10% of another sugar (e.g. glucose or honey): the interaction of molecules of different sugars interfere with the crystallization
- Add an acid ingredient (e.g. lemon juice or cream of tartar) to the sugar
Glucose is perfect to prevent crystallization and is a key ingredient in gummy candies that have to remain soft and not too sweet.
Solubility
Sugars are hydrophilic, meaning they love water and will melt in it. Nothing new for all of you, I suppose. What is important to know is that not all sugars melt the same way in water (i.e. they have a different solubility). The sugar with the highest solubility is fructose, followed by sucrose, glucose, maltose and finally lactose.
As an example, in 100g of water you can melt 69g of glucose, 204g of sucrose or 375g of fructose.